ISO 9001 Revision explained in simple terms
Changes
For September, we are expecting the publication of the two Standards, ISO 9001:2015 and ISO 14001:2015. The FDIS (Final Draft International Standards) are obtainable at the Austrian Standards Institute (ISO/FDIS 9001:2015, ISO/FDIS 14001:2015).
In a series of technical lectures, Quality Austria provides information on the revision of ISO 9001:2015. Each month, a key concept of the revision will be explained more profoundly. This month, Wolfgang Pölz, MSc, MBA, explains the requirements relating to the topic of changes and the way to implement them.
Requirements relating to the topic of changes
Wolfgang Pölz, MSc, MBA
Effectively managing changes - a key issue of the ISO 9001:2015 Revision. Organizations are confronted with environments that are increasingly becoming dynamic and complex. In order to be capable of fulfilling the customer expectations in these environments, it will often be necessary to use different forms of the further development of a system. This will also include corrective action and continual improvement, e.g. trailblazing change, innovation or re-structuring.
A quality management system certified acc. to ISO 9001:2015 is required to have a clear systematics enabling an organization to develop further and to handle changes of products and services. The most important requirements relating to this topic are the following:
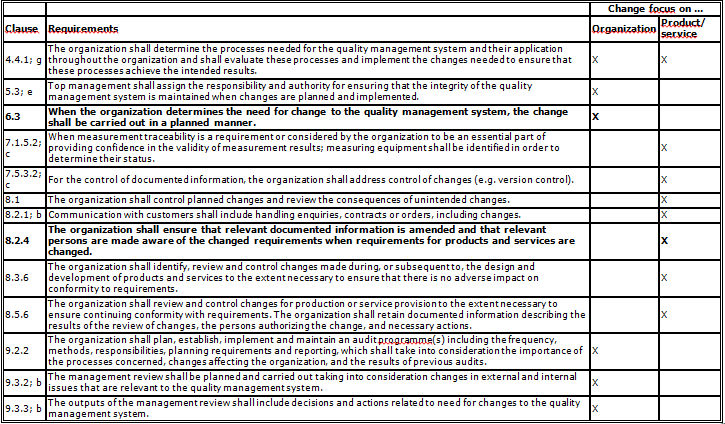
Table 1: Mentions of „change“ in ISO 9001:2015
Clauses 6.3 (Planning of changes) and 8.2.4 (Changes to requirements for products and services) present fundamental requirements to be taken into account for the other Clauses mentioned here.
Clause 6.3 Planning of changes explains what needs to be considered when changing the quality management system. It will be necessary to consider four aspects, which are shown in Figure 1:
Figure 1: Changes to the quality management system
(a) On the hand, the purpose and consequence of the changes are focused on creating clear value - why is this changed made, and what exactly is to be achieved by making this change? On the other hand, both risk-based thinking and the topic of interactions (see ISO 9001:2015; 4.4) are addressed explicitly.
(b) Integrity of the quality management system requires organizations to make changes as to make sure that the quality management system that works well and the ability to deliver products conforming to the requirements and/or to provide conforming services are not put at risk.
(c) Availability of resources refers to tangible and human resources (incl. competence!). The latter do not only imply the necessity to have a look at the required number of persons but also at their competence for the changed requirements.
(d) Responsibility and authority implies the necessity to review the effects on the structure.
These requirements include central topics of project management to be used in case of changes (change management).
As for change management, the connection between “soft facts” and “hard facts” (“leading change and managing change”) is one of the critical success factors. The vision, incentives and partly ability can also be seen as the “HOW” in terms of leadership and the way to handle it. Thus they are much more significant than the “WITH WHAT”, i.e. the tools and methods used, e.g. resources and action plan.
Eight steps towards the success of change:
(cf. Kotter; Leading Change; S.21ff)
It is true that the steps shown in Figure 2 are separated graphically. In practice, however, these steps will mostly merge, Even if the technical component of change has been elaborated in a well-founded manner, this does not really mean that the employees can handle this change and will ultimately enable its sustainable implementation. People within the organization also need to be ready for this emotionally.
Figure 2: Eight steps towards the success of change; source: cf. Kotter; Leading Change; pp. 21ff
In this context, Gerald Hüther talks about using a “supportive leadership style”, i.e. about a task making it necessary to interlink the know-how existing within the organization, create a positive failure culture, provide for positive experiences and, last but not least, generate regular challenges (cf. cultural change in companies and organizations: Wie hirngerechte Führung funktioniert (The way leadership compatible with the brain works) (access: June 28, 2015). These topics can also be found in a similar form at “leadership” in ISO 9001.
Changes of requirements for products and services
As compared to Clause 7.2.2 of ISO 9001:2008, the requirements have hardly been changed. For purposes of implementation, however, the organization should also develop a system for helping to identify change requests made by the customer: This system, (which may consist in using a checklist, template, etc.), is to make it possible to ensure that all information necessary to evaluate the effects of the change is acquired and evaluated while possibly eliminating unclear points. Thus this approach also significantly supports the organization in meeting the new requirements of Clause 8.5.6 (Control of changes). Usually an integral part of this workflow also consists in ensuring that the adaptation of relevant documented information is secured and that awareness of the persons concerned is raised and that these persons are informed accordingly - for example in the form of changes to the quality management plan. For the cases where non-scheduled changes occur, additional requirements can be found in Clause 8.7 of ISO 9001:2015 (Control of nonconforming outputs).

